Abstract
Zinc (Zn) stable isotope environmental geochemistry is a relatively new field of research that focuses on understanding Zn's geochemical and biogeochemical cycling in natural systems, using Zn isotopic fractionation as a tracer for various processes. Zn isotopes are sensitive to various environmental conditions, including changes in oxidation states, biological uptake, and mineral weathering. This makes them valuable tools for studying both natural and anthropogenic influences on the environment. Zn is one of the most widely used metals in industry and is an essential metal in organic metabolic processes (including those in humans); it is toxic at high concentrations. Zn isotopes have been used to trace sources of contamination in soils and waters, especially in areas affected by mining activities and industrial emissions, thus supporting environmental geochemical assessments and remediation strategies. Zn stable isotope geochemistry has implications on different areas of geosciences, biochemical sciences, and even on medical sciences. This paper provides general information on natural sources of Zn, some of its industrial uses, Zn as an essential element in organic metabolism, and its toxicity. The main focus is to define some isotopic fractionation processes, explain their applications in a general way, define the sample preparation methodologies in the
laboratory, as well as the modern analytical methods to measure Zn isotopic fractionation, and finally, present some preliminary data of Zn isotopic measurements from current projects at Instituto de Geociencias-UNAM, Campus Juriquilla.
References
Adrie, V. y Bert, H. (2002). Sources of Cd, Cu, Pb and Zn in biowaste. Science of the Total Environment, 300, 87-98. https://doi.org/10.1016/S0048-9697(01)01103-2.
Albarède, F. (2004). The stable isotope geochemistry of copper and zinc. Reviews in Mineralogy and Geochemistry, 55 (1), 409–427. https://doi.org/10.2138/gsrmg.55.1.409
Black, J.R., Kavner, A. y Schauble, E.A. (2011). Calculation of equilibrium stable isotope partition function ratios for aqueous zinc complexes and metallic zinc: Geochimica et Cosmochimica Acta, 75(3), 769-783.
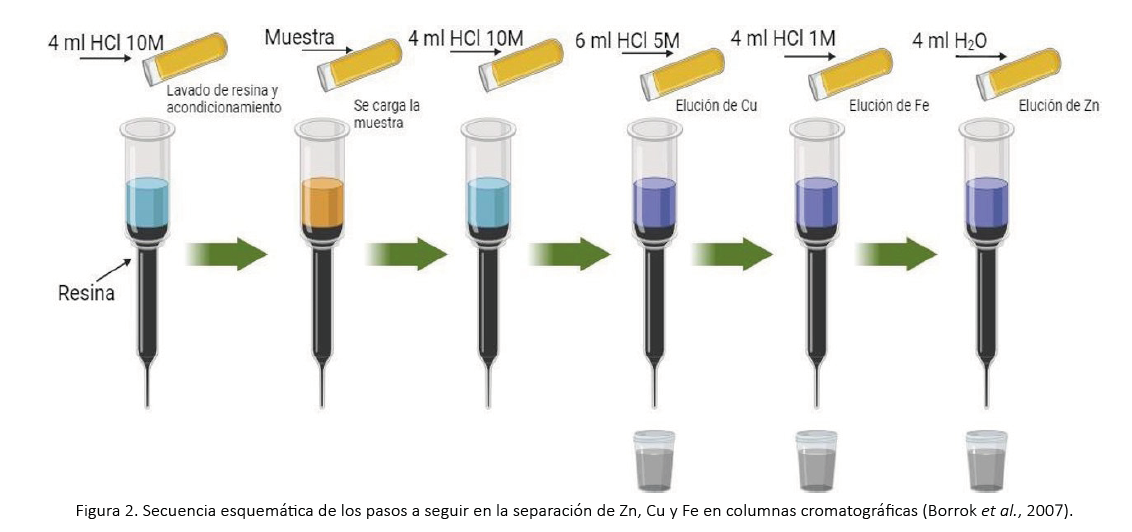
Borrok, D.M., Wanty, R.B., Ridley, W.I., Wolf, R., Lamothe, P.J., y Adams, M. (2007).Separation of copper, iron, and zinc from complex aqueous solutions for isotopic measurement. Chemical Geology, 242 (3-4), 400-414.
Bradl, H. (Ed.). (2005). Heavy metals in the environment: origin, interaction and remediation. Elsevier.
Calvo-Ramos, D., Pribil, M., García, R., Muñoz-Torres, C. y Carrillo- Chávez, A. (2023). Isótopos estables de zinc y su uso como trazadores en el medio ambiente de nieve de alta montaña en México (Iztaccíhuatl y Pico de Orizaba) en Libro de Resúmenes. Convención Geológica Nacional. Sociedad Geológica Mexicana.
Carrillo-Chávez, A., Delgado-Granados H., Vázquez-Selem, L., Ontiveros-González, G., Cortés-Ramos, J., Soto V., Muños-Torres, C. y Calvo-Ramos D. K. (2024). Glaciological studies in Mexico, 60 years of academic work: A summary. Revista Mexicana de Ciencias Geológicas. 41(1), 53–65. https://doi.org/10.22201/cgeo.20072902e.2024.1.1706.
Chai, L., Li, H., Yang, Z., Min, X., Liao, Q., Liu, Y., Men, S., Yan, Y., y Xu, J. (2017). Heavy metals and metalloids in the surface sediments of the Xiangjiang River, Hunan, China: distribution, contamination, and ecological risk assessment. Environmental Science and Pollution Research, 24, 874-885. https://doi.org/10.1007/s11356-016-7872-x
Chen, S., Liu, Y., Hu, J., Zhang, Z., Hou, Z., Huang, F., y Yu, H. (2016). Zinc Isotopic compositions of NIST SRM 683 and wholerock reference materials. Geostandards and Geoanalytical Research, 40(3), 417–432. https://doi.org/10.1111/j.1751-908X.2015.00377.x
Cloquet, C., Carignan, J., Lehmann, M. y Vanhaecke, F. (2007). Variation in the isotopic composition of zinc in the natural environment and the use of zinc isotopes in biogeosciences: a review. Analytical and Bioanalytical Chemistry, 390:451–463, DOI: 10.1007/s00216-007-1635
Druce, M., Stirling, C. H., y Rolison, J. M. (2020). High-precision zinc isotopic measurement of certified reference materials relevant to the environmental, Earth, planetary and biomedical sciences. Geostandards and Geoanalytical Research, 44(4), 711–732. https://doi.org/10.1111/ggr.12341
Environmental Protection Agency (1996). Method 3050B: acid digestion of sediments, sludges and soils, Revision 2. https://www.epa.gov/esam/epa-method-3050b-acid-digestionsediments-sludges-and-soils.
Hou, D., He, J., Lü, C., Ren, L., Fan, Q., Wang, J., y Xie, Z. (2013). Distribution characteristics and potential ecological risk assessment of heavy metals (Cu, Pb, Zn, Cd) in water and sediments from Lake Dalinouer, China. Ecotoxicology and Environmental Safety, 93, 135-144. https://doi.org/10.1016/j.ecoenv.2013.03.012.
John, S.G., Park, J.G., Zhang, Z., y Boyle, E.A., (2007). The isotopic composition of some common forms of anthropogenic zinc. Chemical Geology, 245, 61-69.
John, S.G., Rouxel, O.J., Craddock, P.R., Engwall, A.M. y Boyle, E.A., (2008). Zinc stable isotopes in seafloor hydrothermal vent fluids and chimneys. Earth and Planetary Science Letters, 269, 17-28.
Khodeir, M., Shamy M., Alghamdi M., Zhong M., Sun H., Costa M., Chen L. y Maciejcczyk P. (2012). Source apportionment and elemental composition of PM2. 5 and PM10 in Jeddah City, Saudi Arabia. Atmospheric Pollution Research, 3(3), 331-340. https://doi.org/10.5094/apr.2012.037.
Maret, W. y Sandstead, H. H. (2006). Zinc requirements and the risks and benefits of zinc supplementation. Journal of Trace Elements in Medicine and Biology, 20(1), 3–18. https://doi.org/10.1016/j.jtemb.2006.01.006
Molina, M. J. y Molina, L. T. (2004). Megacities and atmospheric pollution. Journal of the Air & Waste Management Association, 54(6), 644-680. https://doi.org/10.1080/10473289.2004.10470936.
Roohani, N., Hurrell, R., Kelishadi, R., y Schulin, R. (2013). Zinc and its importance for human health : an integrative review. Journal of Research in Medical Sciences, 18, 144–157. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3724376/
Sivry, Y., Riotte, J., Sonke, J.E., Audry S., Schafer, J., Viers, J., Blanc, G., Freydier, R. y Dupre, B., (2008), Zn isotopes as tracers of anthropogenic pollution from Zn-ore smelters The Riou Mort-Lot River system. Chemical Geology, 255, 295-304.
World Health Organization (2022a). Zinc fact sheet for consumers. National Institutes of Health, Office of Dietary Supplements. https://doi.org/10.4324/9780203426968_chapter_1
World Health Organization (2022b). Zinc fact sheet for health professionals. National Institute of Health, Office of Dietary Supplements. https://ods.od.nih.gov/factsheets/Zinc-HealthProfessional/

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright (c) 2024 Universidad Nacional Autónoma de México